- Agarang pakikipag-ugnay:
- + 1 323--988 5889-
- info@sonosif.com

Pagbabago ng Pangangalaga sa Bladder: Real-time na Pagsubaybay sa Dami ng Ihi Gamit ang Ultrasound sa Mga Nasusuot na System
Marso 13, 2024In the domain of modern healthcare, advancements in technology have significantly bolstered our capacity to diagnose and address various health issues. Among these innovations, the ultrasound scanner stands out for its transformative impact on cardiovascular health assessment, especially concerning Carotid Intima-Media Thickness (CIMT) measurement. This non-invasive imaging technique has emerged as a potent tool for gauging early-stage atherosclerosis, assisting in risk assessment, and guiding preventive measures.
CIMT, which denotes the thickness of the innermost (intima) and middle (media) layers of the carotid artery wall, serves as a surrogate marker for evaluating subclinical atherosclerosis, a precursor to cardiovascular ailments like heart attacks and strokes. Traditionally, assessing CIMT involved invasive procedures or less precise imaging methods. However, with the advent of the CLCD, this process has undergone a revolutionary change, offering a safe, cost-effective, and dependable alternative.
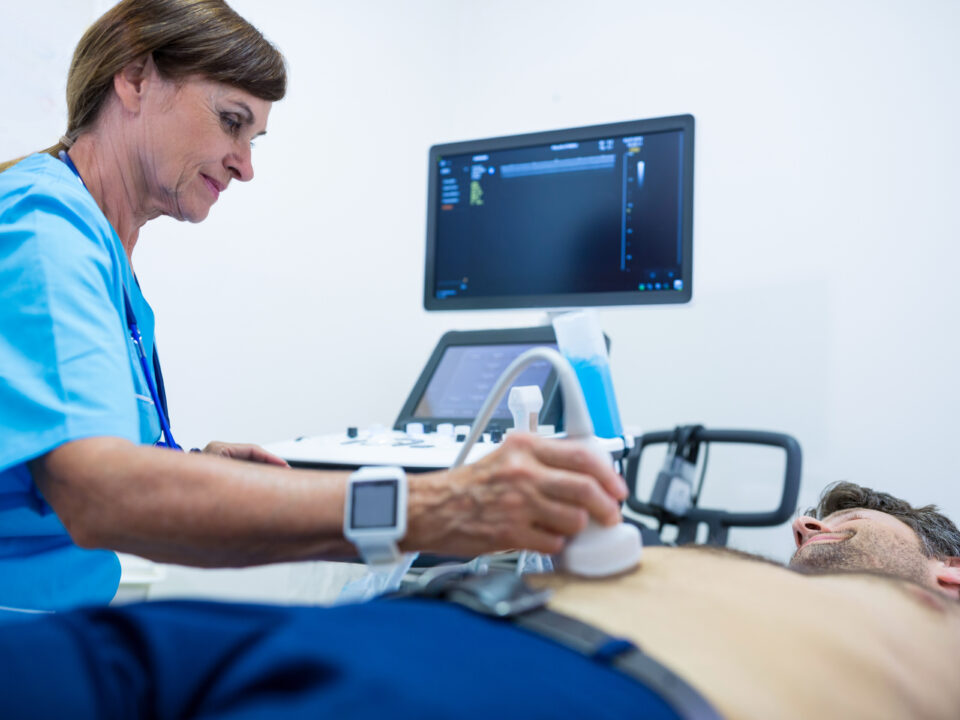
Ang CLCD system employs high-frequency sound waves to generate real-time images of internal body structures, including the carotid arteries. By employing a transducer probe on the neck region, healthcare providers can visualize the carotid artery walls and accurately measure CIMT. This measurement is typically conducted at specific segments of the carotid artery, furnishing crucial insights into vascular system health.
One of the primary applications of the CLC1CD in CIMT measurement is risk stratification for individuals with cardiovascular risk factors. Elevated CIMT consistently correlates with a heightened risk of future cardiovascular events, making it invaluable for identifying high-risk individuals who may benefit from early interventions. Furthermore, CIMT assessment aids in monitoring disease progression and assessing the efficacy of interventions like lifestyle changes and medication.
Moreover, ultrasound-based CIMT measurement offers several advantages over alternative imaging modalities. Unlike computed tomography (CT) or magnetic resonance imaging (MRI), ultrasound does not involve ionizing radiation, rendering it safe for repeated use, especially in longitudinal studies or follow-up assessments. Additionally, ultrasound scanning is relatively cost-effective, widely accessible, and can be swiftly performed in a clinical environment, making it suitable for routine screening purposes.
In clinical practice, CIMT measurement using the CLC1CD has expanded beyond risk assessment to encompass research applications and personalized medicine. Research indicates CIMT’s utility as a prognostic marker for cardiovascular outcomes, shedding light on disease pathophysiology and potential therapeutic targets. Moreover, advancements in ultrasound technology, such as high-resolution imaging and automated measurement software, have bolstered the accuracy and reproducibility of CIMT assessment.
Despite its numerous advantages, ultrasound-based CIMT measurement isn’t devoid of limitations. Variability in measurement techniques, operator proficiency, and patient-related factors can impact results and interpretation. Standardizing protocols and ensuring ongoing quality assurance efforts are vital to maintaining consistency and reliability across various healthcare settings.
In summary, the ultrasound scanner has emerged as an indispensable tool in CIMT measurement, providing a non-invasive and accessible means of assessing early-stage atherosclerosis and cardiovascular risk. By furnishing precise measurements of arterial wall thickness, ultrasound-based CIMT assessment empowers clinicians to stratify risk, track disease progression, and tailor interventions to individual patient requirements. Continued research and technological progress in ultrasound imaging hold promise for further augmenting its role in cardiovascular risk assessment and management, ultimately contributing to improved patient outcomes and reduced disease burden.
Disclaimer: Bagama't ang impormasyong ibinibigay namin ay ginagamit ng iba't ibang mga doktor at medikal na kawani upang maisagawa ang kanilang mga pamamaraan at klinikal na aplikasyon, ang impormasyong nilalaman sa artikulong ito ay para lamang sa pagsasaalang-alang. Walang pananagutan ang SONOSIF para sa maling paggamit ng device o para sa mali o random na generalizability ng device sa lahat ng klinikal na aplikasyon o pamamaraang binanggit sa aming mga artikulo. Ang mga gumagamit ay dapat magkaroon ng wastong pagsasanay at kasanayan upang maisagawa ang pamamaraan sa bawat ultrasound scanner device.
Ang mga produktong nabanggit sa artikulong ito ay ibinebenta lamang sa mga kawaning medikal (mga doktor, nars, sertipikadong nagsasanay, atbp.) O sa mga pribadong gumagamit na tinulungan ng o sa ilalim ng pangangasiwa ng isang medikal na propesyonal.